UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported): January 13, 2020
(Exact name of registrant as specified in its charter)
(State or other jurisdiction | (Commission File Number) | (IRS Employer | ||
of incorporation) | Identification No.) | |||
(Address of principal executive offices, including zip code)
(240 ) 631-3200
(Registrant’s telephone number, including area code)
N/A
(Former name or former address, if changed since last report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions (see General Instruction A.2. below):
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company ☐
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Securities registered pursuant to Section 12(b) of the Act:
Title of each class | Trading Symbol(s) | Name of each exchange on which registered |
Item 2.02 Results of Operations and Financial Condition.
On January 13, 2020, Emergent BioSolutions Inc. announced preliminary unaudited financial results for 2019 and guidance for 2020. The full text of the press release issued in connection with the announcement is furnished as Exhibit 99.1 to this Current Report on Form 8-K. In addition, the sections entitled “Who we are today,” “Consistent diversified revenue growth…,” “…Driving strong profitability,” “2019 financial goals,” “Reconciliation of Net Income to Adjusted Net Income” and “Reconciliation of Net Income to EBITDA and Adjusted EBITDA” of the corporate slide deck furnished as Exhibit 99.2 are incorporated herein by reference.
Item 7.01. Regulation FD Disclosure
During the week of January 13, 2020, representatives of the company will be attending meetings with investors, analysts and others at the J.P. Morgan Healthcare Conference in San Francisco, California and these company representatives will present the slides attached as Exhibit 99.2 to this Current Report on Form 8-K.
Item 9.01 Financial Statements and Exhibits.
(d) Exhibits.
Exhibit No. | Description | |
99.1 | ||
99.2 | ||
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
EMERGENT BIOSOLUTIONS INC. | ||
Dated: January 13, 2020 | By: | /s/ RICHARD S. LINDAHL |
Name: Richard S. Lindahl Title: Executive Vice President, Chief Financial Officer and Treasurer | ||


EMERGENT BIOSOLUTIONS ANNOUNCES PRELIMINARY 2019 FINANCIAL RESULTS AND PROVIDES 2020 FINANCIAL FORECAST
• | Full year 2019 preliminary revenues and adjusted profitability metrics in line with prior guidance |
• | Full year 2020 forecast reflects continued growth of core business as well as investments in key development programs and operational capabilities |
GAITHERSBURG, Md., January 13, 2020—Emergent BioSolutions Inc. (NYSE: EBS) today announced selected preliminary unaudited 2019 financial results and its financial forecast for 2020.
Robert G. Kramer Sr., president and chief executive officer of Emergent BioSolutions Inc. stated, “Our preliminary results for 2019 reflect another strong year of execution on the part of the entire Emergent team. With total revenue of $1.1 billion, anticipated gross margin of 56% to 58%, which is a 200 to 400 basis point improvement over prior year, and margins on adjusted net income and adjusted EBITDA of 14% (1) and 26% (2), respectively, our financial performance reflects the consistency and durability of our business model and its focus on public health threat solutions. Moreover, we achieved our key operational goals for the year, which included securing over $3 billion of renewed contract value, expanding markets for our existing products and services portfolio, and continuing investments to advance our product pipeline and enhance our manufacturing infrastructure.”
Continuing, Kramer stated, “As we enter 2020, we are planning for continued expansion of our business and a financial forecast that at the midpoint anticipates 11% year-over-year growth in total revenue along with strong absolute and relative gains in adjusted net income and adjusted EBITDA. We intend to achieve these outcomes while simultaneously expanding our portfolio of advanced stage product candidates that address serious global public health threats. As a result, we are confident in the strength of our business model as we pursue the sustained growth and value creation to be realized in the 2020-2024 growth strategy we announced at the Analyst & Investor Day we held in November, which contemplates annual revenue in excess of $2 billion by 2024.”
PRELIMINARY 2019 FINANCIAL RESULTS (Unaudited)
The company is providing the following preliminary, unaudited financial results for full year 2019.
(in millions) | PRELIMINARY RESULTS (As of 1/13/2020) | PRIOR GUIDANCE (As of 11/6/2019) |
Total Revenues | $1,100 -- $1,105 | $1,060 -- $1,140 |
Net Income | $53 -- $63 | $80 -- $110 |
Adjusted Net Income (3) | $150 -- $160 | $150 -- $180 |
EBITDA (3) | $225 -- $235 | $250 -- $280 |
Adjusted EBITDA (3) | $280 -- $290 | $280 -- $310 |
Total Revenue
For the full year 2019, the company anticipates total revenue of $1,100 to $1,105 million, the midpoint of which represents a $320 million or 41% increase from 2018, the second year in a row of approximately 40% year-over-year growth in total revenue. This annual increase is due primarily to the contribution of sales of NARCAN® (naloxone HCl) Nasal Spray and ACAM2000® (Smallpox (Vaccinia) Vaccine, Live) in 2019 as well as higher contracts and grants revenue, offset by lower anthrax vaccine revenue (now a combination of BioThrax® (Anthrax Vaccine Adsorbed) and AV7909 (Anthrax Vaccine Adsorbed, Adjuvanted) (4)).
1
Net Income (GAAP and Adjusted)
For the full year 2019, the company anticipates net income of $53 to $63 million, which reflects the impact of two significant items, contingent consideration related to the Adapt acquisition and a non-cash impairment. The company also anticipates adjusted net income of $150 to $160 million, the midpoint of which represents a $33 million or 27% increase from 2018, reflecting the impact of both increased product sales as well as higher average margins on the products contributing to product sales in 2019. (See “Reconciliation of Net Income to Adjusted Net Income, EBITDA and Adjusted EBITDA” for a definition of terms and reconciliation tables.)
Note:
The preliminary 2019 financial results are subject to revision and will be finalized upon completion of the company’s external audit, which is anticipated by late February 2020. The company's final audited financial results could differ materially from these selected preliminary results.
2020 FINANCIAL FORECAST
(in millions) | FULL YEAR 2020 (As of 1/13/2020) |
Total Revenues | $1,175 -- $1,275 |
Adjusted Net Income (3) | $160 -- $210 |
Adjusted EBITDA (3) | $300 -- $360 |
For the full year of 2020, the company’s financial forecast includes the impact of the following items:
• | continued growth in sales of NARCAN Nasal Spray to a range of $285 -- $315 million; |
• | combined deliveries of AV7909 (4) and BioThrax to the Strategic National Stockpile (SNS) in a range of $270 -- $300 million; |
• | deliveries of ACAM2000 in a range of $180 -- $200 million under procurement contracts with the U.S. government and other foreign governments; |
• | deliveries of raxibacumab to the SNS under the anticipated follow-on procurement contract with the ASPR; |
• | domestic and international sales of the other medical countermeasures that comprise Other Product sales; |
• | continued expansion of CMO services revenue; |
• | continued improvement of gross margin in a range of 200 -- 400 basis points, driven by improved product mix; and |
• | continued investment in discretionary development projects funded by the company, most notably the anticipated Phase 3 studies for both the Chikungunya and FLU-IGIV product candidates, among other R&D projects. |
The outlook for 2020 does not include estimates for potential new corporate development or other M&A transactions.
PRESENTATION WEBCAST
The company will provide an update on the current business and discuss preliminary 2019 financial results, the forecast and corporate goals for 2020, and long-term goals for the five-year period 2020-2024 during its presentation at the 38th Annual J.P. Morgan Healthcare Conference on January 14, 2020 at 10:30 AM Pacific time.
A live webcast of the presentation can be accessed through Emergent’s website. Visit www.emergentbiosolutions.com and select the “Investors” section. An on-demand replay of the webcast can also be accessed in the investors section after the presentation has concluded.
FOOTNOTES
(1) | Adjusted Net Income margin is defined as Adjusted Net Income divided by total revenues. For the 2019 Preliminary Results, we reference this metric using the midpoints of the relevant factors. Specifically: $155/$1,103 = 14%. |
(2) | Adjusted EBITDA margin is defined as Adjusted EBITDA divided by total revenues. For the 2019 Preliminary Results, we reference this metric using the midpoints of the relevant factors. Specifically: $285/$1,103 = 26%. |
(3) | See “Reconciliation of Net Income to Adjusted Net Income, EBITDA and Adjusted EBITDA” for a definition of terms and a reconciliation tables. |
(4) | AV7909 is a product candidate not yet approved by the FDA or any other health regulatory agency but procured by the U.S. government under special circumstances. |
RECONCILIATION OF NET INCOME TO ADJUSTED NET INCOME, EBITDA AND ADJUSTED EBITDA
This press release contains five financial measures (Adjusted Net Income, Adjusted Net Income margin, EBITDA (Earnings Before Interest, Taxes, Depreciation and Amortization), Adjusted EBITDA and Adjusted EBITDA margin) that are considered “non-GAAP” financial measures under applicable Securities and Exchange Commission rules and regulations. These non-GAAP financial measures should be considered supplemental to and not a substitute for financial information prepared in accordance with generally accepted accounting principles. The Company’s definition of these non-GAAP measures may differ from similarly titled measures used by others. Adjusted Net Income adjusts for specified items that can be highly variable or difficult to predict, or reflect the non-cash impact of charges resulting from purchase accounting. All adjustments are tax effected utilizing the federal statutory tax rate for the US, except for changes in the fair value of contingent consideration as the vast majority is non-deductible for tax purposes. Adjusted Net Income margin is defined as Adjusted Net Income divided by total revenues. EBITDA reflects net income excluding the impact of depreciation, amortization, interest expense and provision for income taxes. Adjusted EBITDA also excludes specified items that can be highly variable and the non-cash impact of certain purchase accounting adjustments. Adjusted EBITDA margin is defined as Adjusted EBITDA divided by total revenues. The Company views these non-GAAP financial measures as a means to facilitate management’s financial and operational decision-making, including evaluation of the Company’s historical operating results and comparison to competitors’ operating results. These non-GAAP financial measures reflect an additional way of viewing aspects of the Company’s operations that, when viewed with GAAP results and the reconciliations to the corresponding GAAP financial measure may provide a more complete understanding of factors and trends affecting the Company’s business.
The determination of the amounts that are excluded from these non-GAAP financial measures are a matter of management judgment and depend upon, among other factors, the nature of the underlying expense or income amounts. Because non-GAAP financial measures exclude the effect of items that will increase or decrease the Company’s reported results of operations, management strongly encourages investors to review the Company’s consolidated financial statements and publicly filed reports in their entirety.
Reconciliation of Net Income to Adjusted Net Income (Unaudited)
($ in millions) | Twelve Months Ended December 31, | |||
2020 (Forecast) | 2019 (Estimated) | 2018 (Actual) | Source | |
Net Income | $105.0 to $155.0 | $53.0 to $63.0 | $62.7 | NA |
Adjustments: | ||||
+ Non-cash amortization charges | 64.0 | 61.0 | 25.9 | SG&A, Other Income |
+ Change in fair value of contingent consideration | 1.0 | 24.0 | 3.1 | COGS |
+ Acquisition-related costs (transaction & integration) | 4.0 | 13.0 | 27.3 | SG&A |
+ IPR&D intangible asset impairment | - | 12.0 | - | R&D |
+ Impact of purchase accounting on inventory step-up | - | 6.0 | 18.4 | COGS |
+ Exit and disposal costs | - | - | 0.4 | SG&A |
Tax effect | (14.0) | (19.0) | (15.1) | NA |
Total Adjustments | 55.0 | 97.0 | 60.0 | NA |
Adjusted Net Income | $160.0 to $210.0 | $150.0 to $160.0 | $122.7 | NA |
3
Reconciliation of Net Income to EBITDA and Adjusted EBITDA (Unaudited)
($ in millions) | Twelve Months Ended December 31, | |||
2020 (Forecast) | 2019 (Estimated) | 2018 (Actual) | Source | |
Net Income | $105.0 to $155.0 | $53.0 to $63.0 | 62.7 | NA |
Adjustments: | ||||
+ Depreciation & Amortization | 111.0 to 121.0 | 111.0 | 61.3 | COGS, SG&A, R&D |
+ Total Interest Expense | 31.0 | 38.0 | 9.9 | Other Expense/ (Income) |
+ Provision for Income Taxes | 48.0 | 23.0 | 18.8 | Income Taxes |
Total Adjustments | 190.0 to 200.0 | 172.0 | 90.0 | NA |
EBITDA | $295.0 to $355.0 | $225.0 to $235.0 | $152.7 | NA |
Additional Adjustments: | ||||
+ Change in fair value of contingent consideration | 1.0 | 24.0 | 3.1 | COGS |
+ Acquisition-related costs (transaction & integration) | 4.0 | 13.0 | 27.3 | SG&A |
+ IPR&D intangible asset impairment | - | 12.0 | - | R&D |
+ Impact of purchase accounting on inventory step-up | - | 6.0 | 18.4 | COGS |
+ Exit and disposal costs | - | - | 0.4 | SG&A |
Total Additional Adjustments | 5.0 | 55.0 | 49.2 | NA |
Adjusted EBITDA | $300.0 to $360.0 | $280.0 to $290.0 | $201.9 | NA |
ABOUT EMERGENT BIOSOLUTIONS INC.
As a global life sciences company whose mission is to protect and enhance life, we provide solutions that target public health threats. Through our specialty products and services as well as our social responsibility efforts, we aspire to build healthier, safer communities and deliver peace of mind to our patients and customers so they can focus on what’s most important in their lives. For more information visit www.emergentbiosolutions.com. Find us on LinkedIn and follow us on Twitter @emergentbiosolu and Instagram @life_at_emergent.
SAFE HARBOR STATEMENT
This press release includes forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Any statements, other than statements of historical fact, including, without limitation, our financial guidance and related projections and statements regarding our ability to meet such projections in the anticipated timeframe, if at all, and statements regarding the continued expansion of our core business, growth of NARCAN Nasal Spray sales, deliveries of ACAM2000 and raxibacumab to the SNS, our entry into the anticipated raxibacumab follow-on procurement contract with ASPR; continued expansion of CMO services revenue; continued gross margin improvement, the results of our continued investment in discretionary development product candidates and clinical trials for both Chikungunya and FLU-IGIV and any other statements containing the words “will,” “believes,” “expects,” “anticipates,” “intends” “plans,” “targets,” “forecasts,” “estimates” and similar expressions in conjunction with, among other things, discussions of the Company's outlook, financial performance or financial condition, financial and operation goals, strategic goals, growth strategy, product sales, government development or procurement contracts or awards, government appropriations, manufacturing capabilities, and the timing of certain regulatory approvals or expenditures are forward-looking statements. These forward-looking statements are based on our current intentions, beliefs and expectations regarding future events. We cannot guarantee that any forward-looking statement will be accurate. Investors should realize that if underlying
4
assumptions prove inaccurate or unknown risks or uncertainties materialize, actual results could differ materially from our expectations. Investors are, therefore, cautioned not to place undue reliance on any forward-looking statement. Any forward-looking statements speak only as of the date of this press release, and, except as required by law, we do not undertake to update any forward-looking statement to reflect new information, events or circumstances.
There are a number of important factors that could cause our actual results to differ materially from those indicated by such forward-looking statements, including the availability of funding and the exercise of options under our anthrax vaccine contracts; appropriations for the procurement of our products; our ability to continue deliveries of AV7909, ACAM and VIGIV to the SNS; our ability to secure Emergency Use Authorization designation and eventual licensure of AV7909 from the FDA within the anticipated timeframe, if at all; availability of funding for our U.S. government grants and contracts; our ability to successfully integrate and develop the operations, products or product candidates, programs, and personnel of any entities, businesses or products that we acquire, including our acquisitions of PaxVax and Adapt Pharma; our ability to complete expected deliveries of anthrax vaccines, BAT and raxibacumab; our ability to establish a multi-year follow-on contract for raxibacumab; our ability to identify and acquire or in-license products or product candidates that satisfy our selection criteria; our ability and the ability of our collaborators to defend underlying patents from infringement by generic naloxone entrants; whether anticipated synergies and benefits from an acquisition or in-license will be realized within expected time periods, if at all; our ability to utilize our manufacturing facilities and expand our capabilities; our ability and the ability of our contractors and suppliers to maintain compliance with Current Good Manufacturing Practices and other regulatory obligations; the results of regulatory inspections; the success of our ongoing and planned development programs; the timing and results of clinical trials; the timing of and our ability to obtain and maintain regulatory approvals for our product candidates; and our commercialization, marketing and manufacturing capabilities and strategy. The foregoing sets forth many, but not all, of the factors that could cause actual results to differ from our expectations in any forward-looking statement. Investors should consider this cautionary statement as well as the risk factors identified in our periodic reports filed with the Securities and Exchange Commission when evaluating our forward-looking statements.
Investor Contact Robert Burrows Vice President, Investor Relations (o) 240/631-3280; (m) 240/413-1917 | Media Contact Lynn Kieffer Vice President, Corporate Communications (o) 240/631-3391 kiefferl@ebsi.com |
5

Corporate Overview 38th Annual J.P. Morgan Healthcare Conference Robert G. Kramer Sr. President and Chief Executive Officer January 14, 2020

Forward-looking statements / Non-GAAP financial measures / trademarks Safe-Harbor Statement This presentation includes forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Any statements, other than statements of historical fact, including, without limitation, our financial forecast and guidance, statements regarding our continued success, becoming a Fortune 500 company, our sustainable business model and competitive advantages, building on scalable capabilities, statements about consistent, diversified growth, profitability, doubling revenues and achieving target adjusted EBITDA margin, covering a larger portion of the public health threat market, growth through M&A, strengthening our R&D portfolio and other key growth areas and related future market opportunities and any other statements containing the words “will,” “believes,” “expects,” “anticipates,” “intends” “plans,” “targets,” “forecasts,” “estimates” and similar expressions in conjunction with, among other things, discussions of the Company's outlook, financial performance or financial condition, financial and operation goals, strategic goals, growth strategy, product sales, government development or procurement contracts or awards, government appropriations, manufacturing capabilities, and the timing of certain regulatory approvals or expenditures are forward-looking statements. These forward-looking statements are based on our current intentions, beliefs and expectations regarding future events. We cannot guarantee that any forward-looking statement will be accurate. Investors should realize that if underlying assumptions prove inaccurate or unknown risks or uncertainties materialize, actual results could differ materially from our expectations. Investors are, therefore, cautioned not to place undue reliance on any forward-looking statement. Any forward-looking statements speak only as of the date of this presentation, and, except as required by law, we do not undertake to update any forward-looking statement to reflect new information, events or circumstances. There are a number of important factors that could cause the Company’s actual results to differ materially from those indicated by such forward-looking statements, including the availability of funding and the exercise of options under our anthrax vaccine contracts; appropriations for the procurement of our products; our ability to continue deliveries of AV7909, ACAM and VIGIV to the SNS; our ability to secure Emergency Use Authorization designation and eventual licensure of AV7909 from the FDA within the anticipated timeframe, if at all; availability of funding for our U.S. government grants and contracts; our ability to successfully integrate and develop the operations, products or product candidates, programs, and personnel of any entities, businesses or products that we acquire, including our acquisitions of PaxVax and Adapt Pharma; our ability to complete expected deliveries of anthrax vaccines, BAT and raxibacumab; our ability to establish a multi-year follow-on contract for raxibacumab; our ability to identify and acquire or in-license products or product candidates that satisfy our selection criteria; our ability and the ability of our collaborators to defend underlying patents from infringement by generic naloxone entrants; whether anticipated synergies and benefits from an acquisition or in-license will be realized within expected time periods, if at all; our ability to utilize our manufacturing facilities and expand our capabilities; our ability and the ability of our contractors and suppliers to maintain compliance with Current Good Manufacturing Practices and other regulatory obligations; the results of regulatory inspections; the success of our ongoing and planned development programs; the timing and results of clinical trials; the timing of and our ability to obtain and maintain regulatory approvals for our product candidates; and our commercialization, marketing and manufacturing capabilities and strategy. The foregoing sets forth many, but not all, of the factors that could cause actual results to differ from our expectations in any forward- looking statement. Investors should consider this cautionary statement as well as the risk factors identified in our periodic reports filed with the Securities and Exchange Commission when evaluating our forward-looking statements. Non-GAAP Financial Measures This presentation contains five financial measures (Adjusted Net Income, Adjusted Net Income margin, EBITDA (Earnings Before Interest, Taxes, Depreciation and Amortization), Adjusted EBITDA and Adjusted EBITDA margin) that are considered “non-GAAP” financial measures under applicable Securities and Exchange Commission rules and regulations. These non-GAAP financial measures should be considered supplemental to and not a substitute for financial information prepared in accordance with generally accepted accounting principles. The Company’s definition of these non-GAAP measures may differ from similarly titled measures used by others. Adjusted Net Income adjusts for specified items that can be highly variable or difficult to predict or reflect the non-cash impact of charges resulting from purchase accounting. Adjusted Net Income margin is defined as Adjusted Net Income divided by total revenues. EBITDA reflects net income excluding the impact of depreciation, amortization, interest expense and provision for income taxes. Adjusted EBITDA also excludes specified items that can be highly variable and the non-cash impact of certain purchase accounting adjustments (which are tax effected utilizing the statutory tax rate for the US). Adjusted EBITDA margin is defined as Adjusted EBITDA divided by total revenues. The Company views these non- GAAP financial measures as a means to facilitate management’s financial and operational decision-making, including evaluation of the Company’s historical operating results and comparison to competitors’ operating results. These non-GAAP financial measures reflect an additional way of viewing aspects of the Company’s operations that, when viewed with GAAP results and the reconciliations to the corresponding GAAP financial measure, may provide a more complete understanding of factors and trends affecting the Company’s business. The determination of the amounts that are excluded from these non-GAAP financial measures are a matter of management judgment and depend upon, among other factors, the nature of the underlying expense or income amounts. Because non-GAAP financial measures exclude the effect of items that will increase or decrease the Company’s reported results of operations, management strongly encourages investors to review the Company’s consolidated financial statements and publicly filed reports in their entirety. For additional on the non-GAAP financial measures noted here, please refer to the Reconciliation Tables provided in the Appendix to this presentation. Trademarks BioThrax® (Anthrax Vaccine Adsorbed), RSDL® (Reactive Skin Decontamination Lotion Kit), BAT® [Botulism Antitoxin Heptavalent (A,B,C,D,E,F,G)-(Equine)], Anthrasil® (Anthrax Immune Globulin Intravenous [human]), CNJ-016® [Vaccinia Immune Globulin Intravenous (Human)], Trobigard® (atropine sulfate, obidoxime chloride), ACAM2000®, (Smallpox (Vaccinia) Vaccine, Live), Vivotif® (Typhoid Vaccine Live Oral Ty21a), Vaxchora® (Cholera Vaccine, Live, Oral), NARCAN® (naloxone HCI) Nasal Spray and any and all Emergent BioSolutions Inc. brands, products, services and feature names, logos and slogans are trademarks or registered trademarks of Emergent BioSolutions Inc. or its subsidiaries in the United States or other countries. All other brands, products, services and feature names or trademarks are the property of their respective owners. 2

Our vision To become a Fortune 500 global life sciences company recognized for protecting and enhancing life, driving innovation and living our values Our focus • CBRNE • Emerging Health Crises • Emerging Infectious Disease • Acute/Emergency Care • Travel Health • CDMO services 3
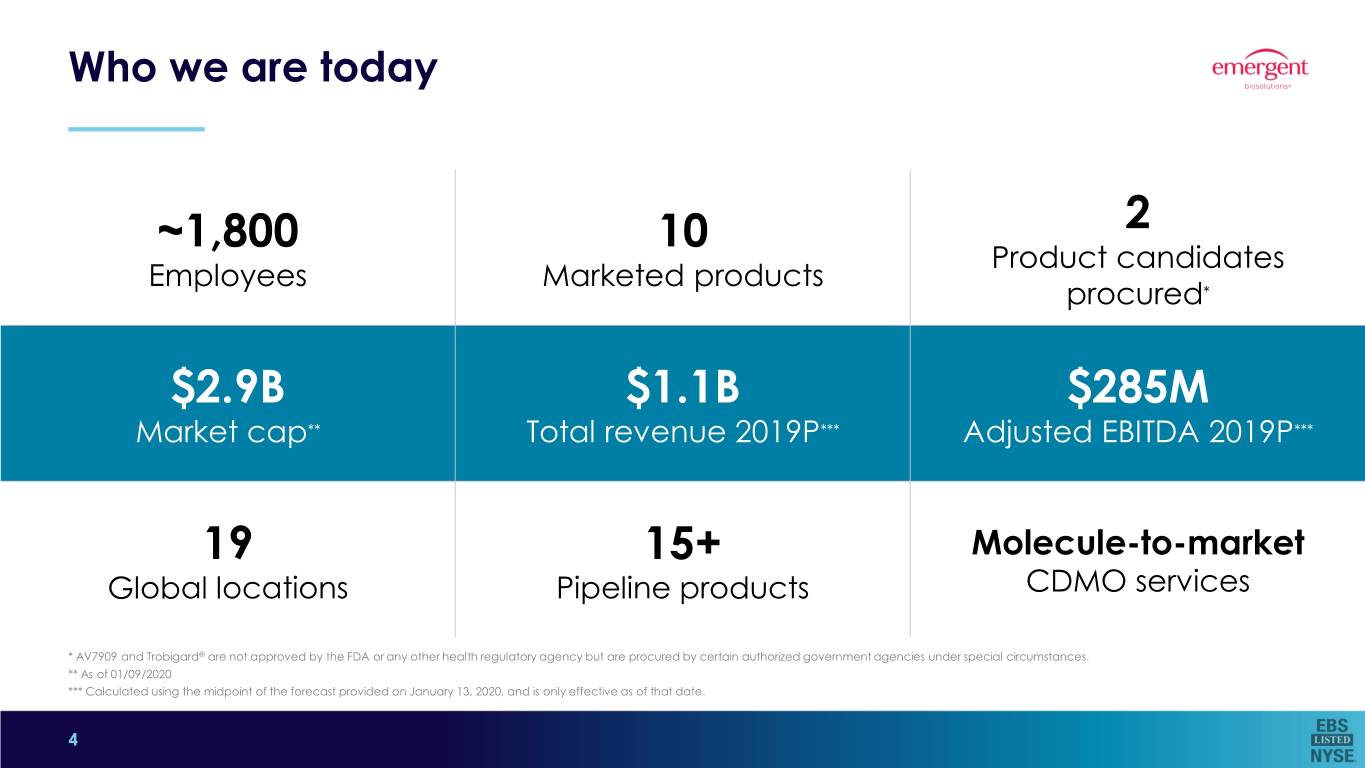
Who we are today ~1,800 10 2 Product candidates Employees Marketed products procured* $2.9B $1.1B $285M Market cap** Total revenue 2019P*** Adjusted EBITDA 2019P*** 19 15+ Molecule-to-market Global locations Pipeline products CDMO services * AV7909 and Trobigard® are not approved by the FDA or any other health regulatory agency but are procured by certain authorized government agencies under special circumstances. ** As of 01/09/2020 *** Calculated using the midpoint of the forecast provided on January 13, 2020, and is only effective as of that date. 4

Our marketed and specially-procured products Product types Anthrax Smallpox Opioids Anthrasil® ACAM2000® NARCAN® [Anthrax Immune (Smallpox (Vaccinia) (naloxone HCl) Globulin Intravenous Vaccine, Live) Nasal Spray (human)] ® Vaccines VIGIV CNJ-016 AV7909* [Vaccinia Immune Public (liquid, oral) [Anthrax Vaccine Globulin Intravenous Chemical Adsorbed (AVA), (Human)] ® Health Adjuvanted] RSDL (Reactive Skin Decontamination Threats ® BioThrax Lotion Kit) (Anthrax Vaccine Travel Health Emerging Therapeutics Adsorbed) ®* EmergingHealth Vaxchora® Trobigard (hyperimmune/mAb) (atropine sulfate, Health (Cholera Vaccine, Crises raxibacumab obidoxime chloride Crises Live, Oral) injection auto-injector) A fully human 5 ® monoclonal antibody Vivotif (Typhoid Vaccine Live Oral Ty21a) Botulism >$30B Market Medical devices (device, drug-device BAT® Opportunity combination product) [Botulism Antitoxin Heptavalent (A, B, C, D, E, F, G) - (Equine)] * AV7909 and Trobigard® are not approved by the FDA or any other health regulatory agency but are procured by certain authorized government agencies under special circumstances. 5

Our services Molecule-to-market Sustainable competitive advantages CDMO offerings • Foundational market approach • Science and technology Technology Development services • Industry-leading track record Platforms • Speed and flexibility to market • Tailored, individualized and integrated offerings Drug substance • 9 Global development & manufacturing sites • Center for Innovation in Advanced Development and Manufacturing (CIADM) $20B Market Opportunity Drug product & packaging 6
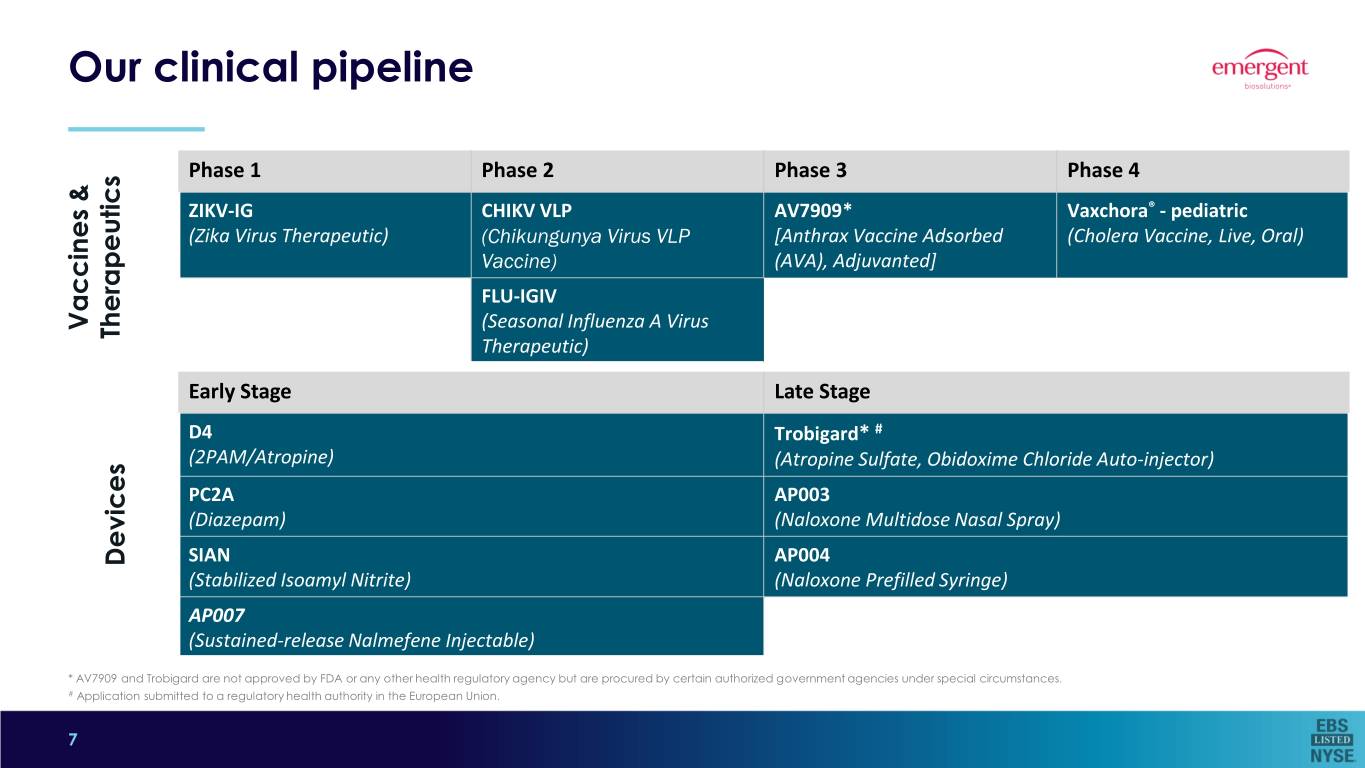
Our clinical pipeline Phase 1 Phase 2 Phase 3 Phase 4 ZIKV-IG CHIKV VLP AV7909* Vaxchora® - pediatric (Zika Virus Therapeutic) (Chikungunya Virus VLP [Anthrax Vaccine Adsorbed (Cholera Vaccine, Live, Oral) Vaccine) (AVA), Adjuvanted] FLU-IGIV Vaccines & Vaccines (Seasonal Influenza A Virus Therapeutics Therapeutic) Early Stage Late Stage D4 Trobigard* # (2PAM/Atropine) (Atropine Sulfate, Obidoxime Chloride Auto-injector) PC2A AP003 (Diazepam) (Naloxone Multidose Nasal Spray) Devices SIAN AP004 (Stabilized Isoamyl Nitrite) (Naloxone Prefilled Syringe) AP007 (Sustained-release Nalmefene Injectable) * AV7909 and Trobigard are not approved by FDA or any other health regulatory agency but are procured by certain authorized government agencies under special circumstances. # Application submitted to a regulatory health authority in the European Union. 7
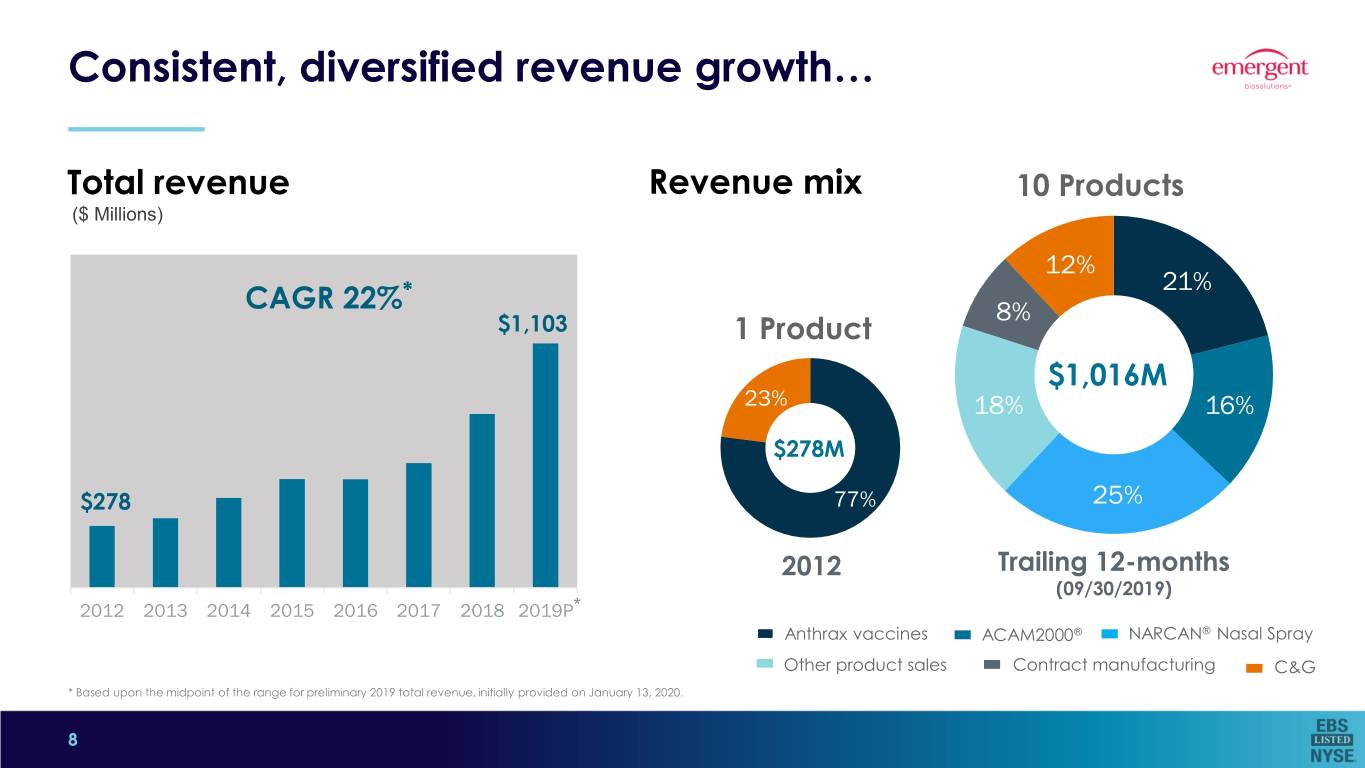
Consistent, diversified revenue growth… Total revenue Revenue mix 10 Products ($ Millions) 12% * 21% CAGR 22% 8% $1,103 1 Product $1,016M 23% 18% 16% $278M $278 77% 25% 2012 Trailing 12-months (09/30/2019) 2012 2013 2014 2015 2016 2017 2018 2019P* Anthrax vaccines ACAM2000® NARCAN® Nasal Spray Other product sales Contract manufacturing C&G * Based upon the midpoint of the range for preliminary 2019 total revenue, initially provided on January 13, 2020. 8
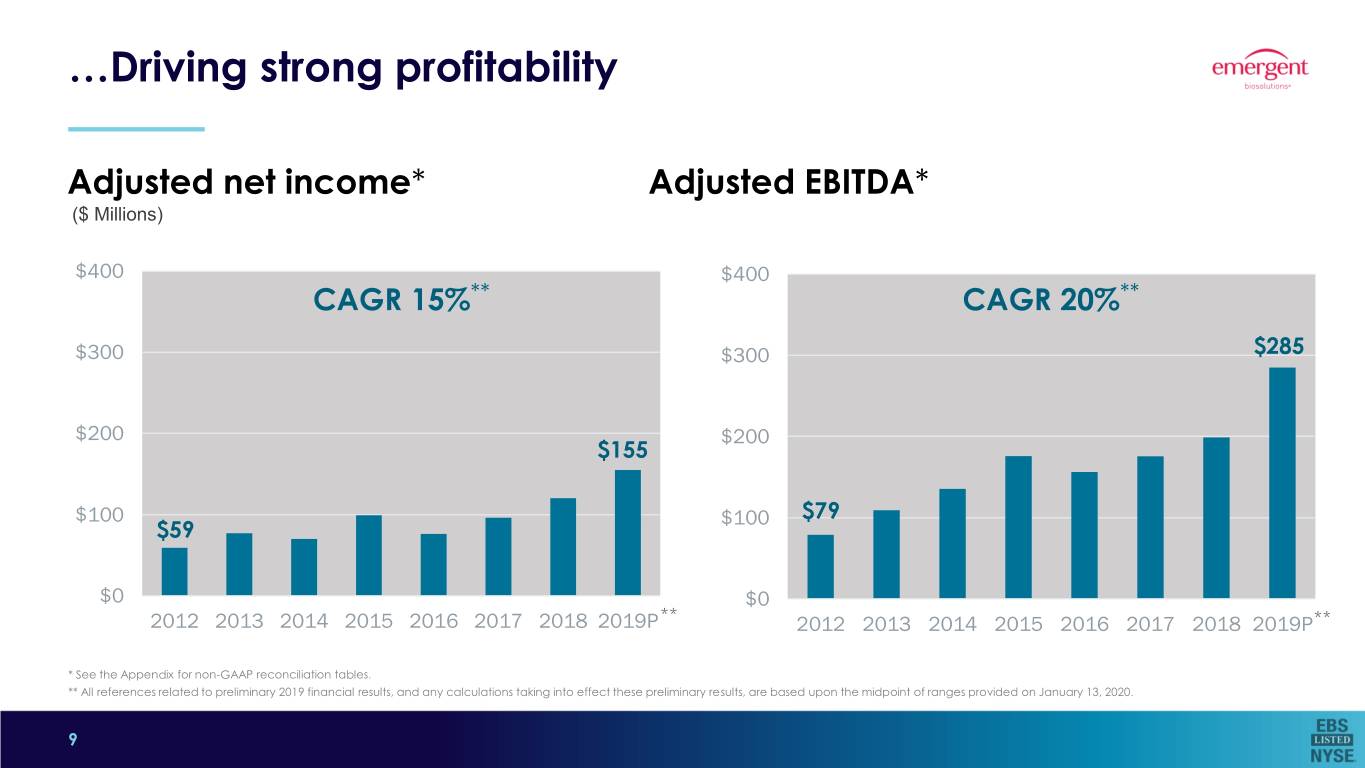
…Driving strong profitability Adjusted net income* Adjusted EBITDA* ($ Millions) $400 $400 CAGR 15%** CAGR 20%** $300 $300 $285 $200 $200 $155 $100 $100 $79 $59 $0 $0 ** 2012 2013 2014 2015 2016 2017 2018 2019P 2012 2013 2014 2015 2016 2017 2018 2019P** * See the Appendix for non-GAAP reconciliation tables. ** All references related to preliminary 2019 financial results, and any calculations taking into effect these preliminary results, are based upon the midpoint of ranges provided on January 13, 2020. 9
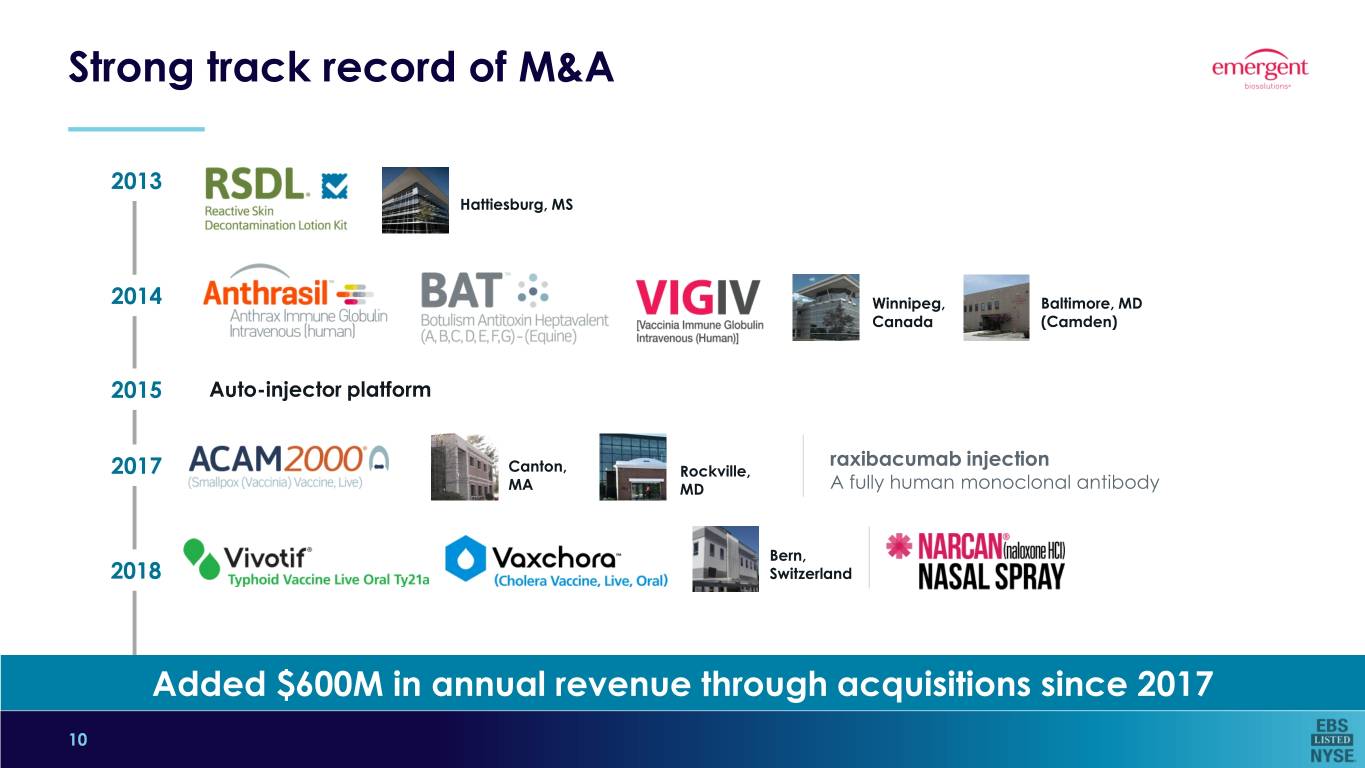
Strong track record of M&A 2013 Hattiesburg, MS 2014 Winnipeg, Baltimore, MD Canada (Camden) 2015 Auto-injector platform raxibacumab injection 2017 Canton, Rockville, MA MD A fully human monoclonal antibody Bern, 2018 Switzerland Added $600M in annual revenue through acquisitions since 2017 10

2019 financial goals Full Year Financial Goals Preliminary Financial Results (As of 1/13/2020) Total Revenue $1,060M-$1,140M $1,100M-$1,105M*** Adjusted Net $150M-$160M*** Income* $150M-$180M # Margin** 15% 14% Adjusted $280M-$290M*** EBITDA* $280M-$310M # Margin** 27% 26% * See the Appendix for non-GAAP reconciliation tables. ** Calculated using the midpoint of the range of the relevant metric divided by the midpoint of the range for total revenue. *** Based upon the ranges provided in the press release issued by the Company on January 13, 2020. # See the Appendix for methodology and specific factors used in calculating this metric. 11

2019 operational goals Operational Goals Operational Achievements • ACAM2000 - $2B – 10 year contract ✓ Secure/renew U.S. Government • VIG - $535M – 10 year contract procurement contracts for key programs • BAT – up to $500M – 10 year contract • AV7909 Exercise - $261M – 1 year contract ✓ Continue initiatives to support awareness, • Expanded annual production capacity to >10M units • Supported implementation of Co-Rx in CA, NM, WA, OH availability and affordability of NARCAN® • Price has remained constant with average copay $19 per Nasal Spray 4 mg two-unit carton • AV7909: P3 initiated; initial doses delivered to SNS • Vaxchora: New formulation (refrigerated versus frozen) ✓ Advance 6 R&D programs to next stage • CHIKV VLP: P2 completed; P3 development plans reviewed of development • FLU-IGIV: P2 database locked; safety analysis ongoing • Naloxone PFS: FDA dossier submission completed • Trobigard: Belgium Health Authority submission completed 12

Title Only 13 Footer

2024 strategic goals • Double revenue to >$2B • Grow in disciplined, profitable way; achieve adjusted EBITDA margin of 27%-30%* • Expand and build scalable leadership positions in current and new public health threat (PHT) markets • Invest in capabilities, innovation and operational excellence * Defined as Adjusted EBITDA divided by total revenue. 14
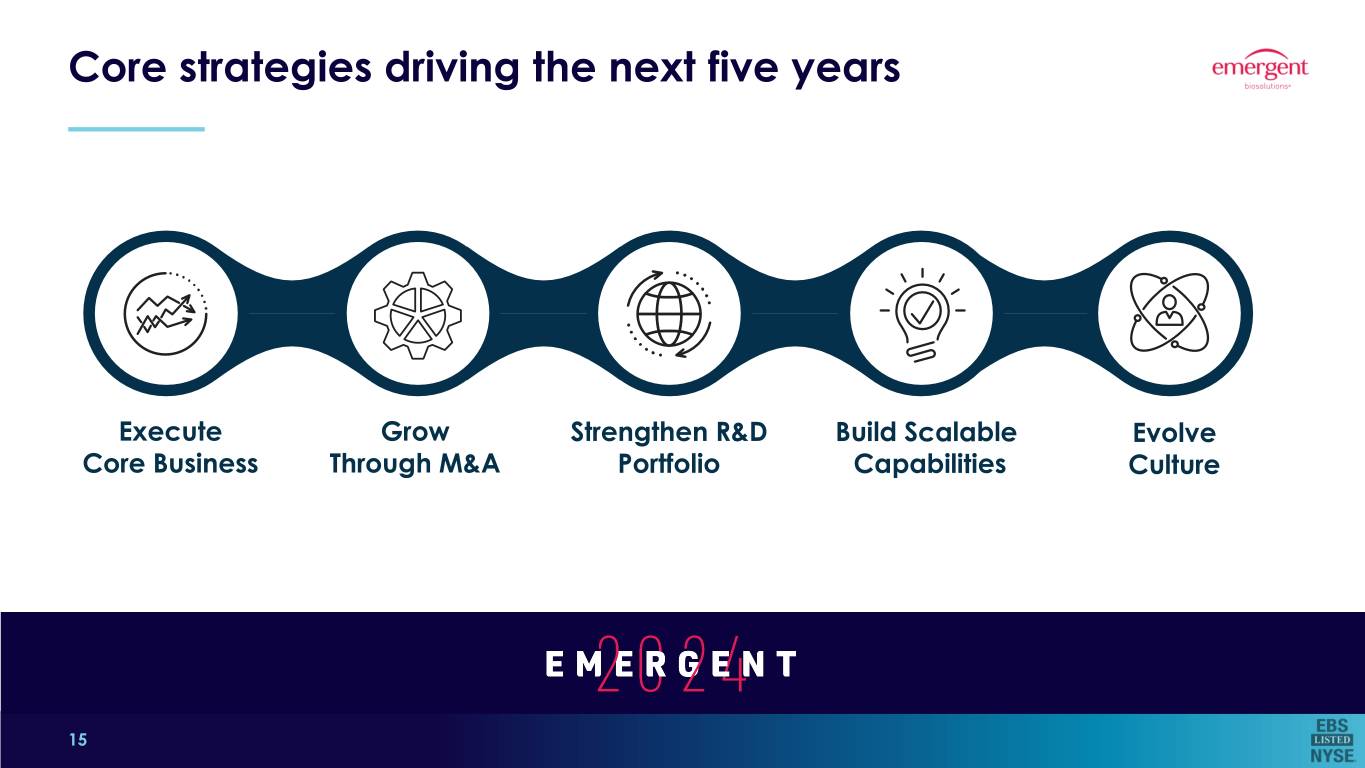
Core strategies driving the next five years Execute Grow Strengthen R&D Build Scalable Evolve Core Business Through M&A Portfolio Capabilities Culture * Defined as Adjusted EBITDA divided by total revenue. 15

Core strategy – Execute Core Business Deliver core business in products and services Execute Core Business * Defined as Adjusted EBITDA divided by total revenue. 16
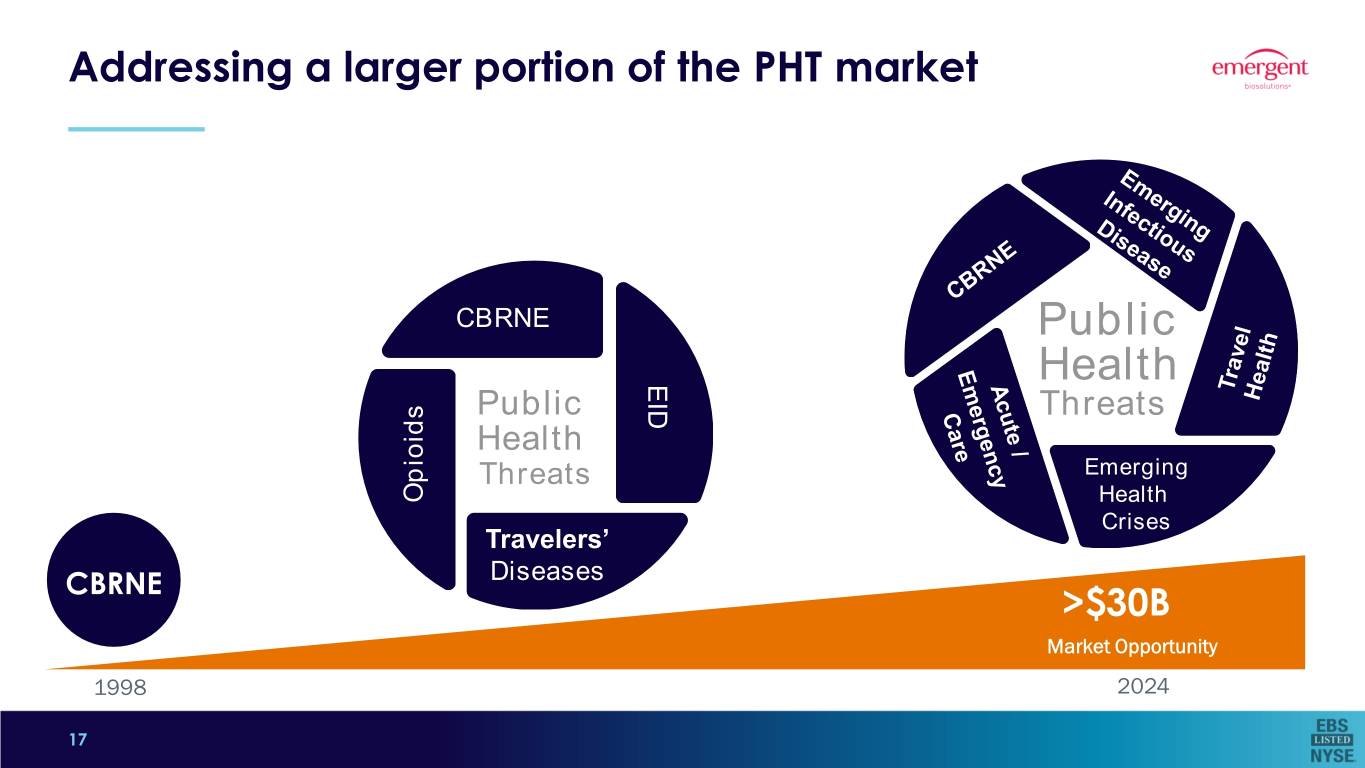
Addressing a larger portion of the PHT market CBRNE Public EID Health Public Threats Health Threats EmergingEmerging Opioids HealthHealth CrisesCrises Travelers’ CBRNE Diseases >$30B Market Opportunity 1998 2024 17

2020-2024 corporate growth strategy targets large addressable market opportunities Public CDMO Health Technology Threats Platform Emerging EmergingHealth HealthCrises Crises 5 >$30B Market Opportunity $20B Market Opportunity 18
Customer and partner mix provides platform for continued success Pharma and NGO Government Clinics/Distributors/Pharmacies Biotech Small Mid Large >50 customers 19

Core strategy – Grow Through M&A Expand impact on patients and customers while profitably delivering incremental topline revenue Grow Through M&A 20

M&A growth driven by a disciplined approach Criteria • Strategic fit Public • Preference for products Health accretive in less than 24 months Threats Emerging • Ability to generate risk-adjusted EmergingHealth HealthCrises returns Crises 5 21

Core strategy – Strengthen R&D Portfolio Build R&D pipeline to become a meaningful contributor to growth after 2024 Strengthen R&D Portfolio 22

Core strategy – Build Scalable Capabilities Invest in operational excellence and innovation to support a growing enterprise that will deliver greater impact Build Scalable Capabilities 23

Core strategy – Evolve Culture Evolve our culture to support increased employee engagement and empowerment Evolve Culture 24

2020 financial forecast Total Revenue Adjusted EBITDA* Adjusted Net Income* $1,175M-$1,275M** $300M-$360M** $160M-$210M** • NARCAN: $285-$315M • Gross Margin Improvement of • Anthrax Vaccines: $270-$300M 200-400 bps. • ACAM2000: $180-$200M Note: 2020 financial forecast does not include impact of M&A. * See the Appendix for non-GAAP reconciliation tables. ** Based upon the ranges provided in the press release issued by the Company on January 13, 2020. 25

Summary takeaways Proven track record – build from history of profitable, diversified revenue growth Scalable and sustainable business model – deliver expanding offering of specialty products and services addressing global preparedness and response Established leader – continue to build and scale leadership positions in select PHT markets where we have competitive advantages Strong financial foundation – employ a disciplined capital deployment approach to support strategic objectives and drive shareholder value 26

Corporate Overview 38th Annual J.P. Morgan Healthcare Conference Robert G. Kramer Sr. President and Chief Executive Officer January 14, 2020

Appendix
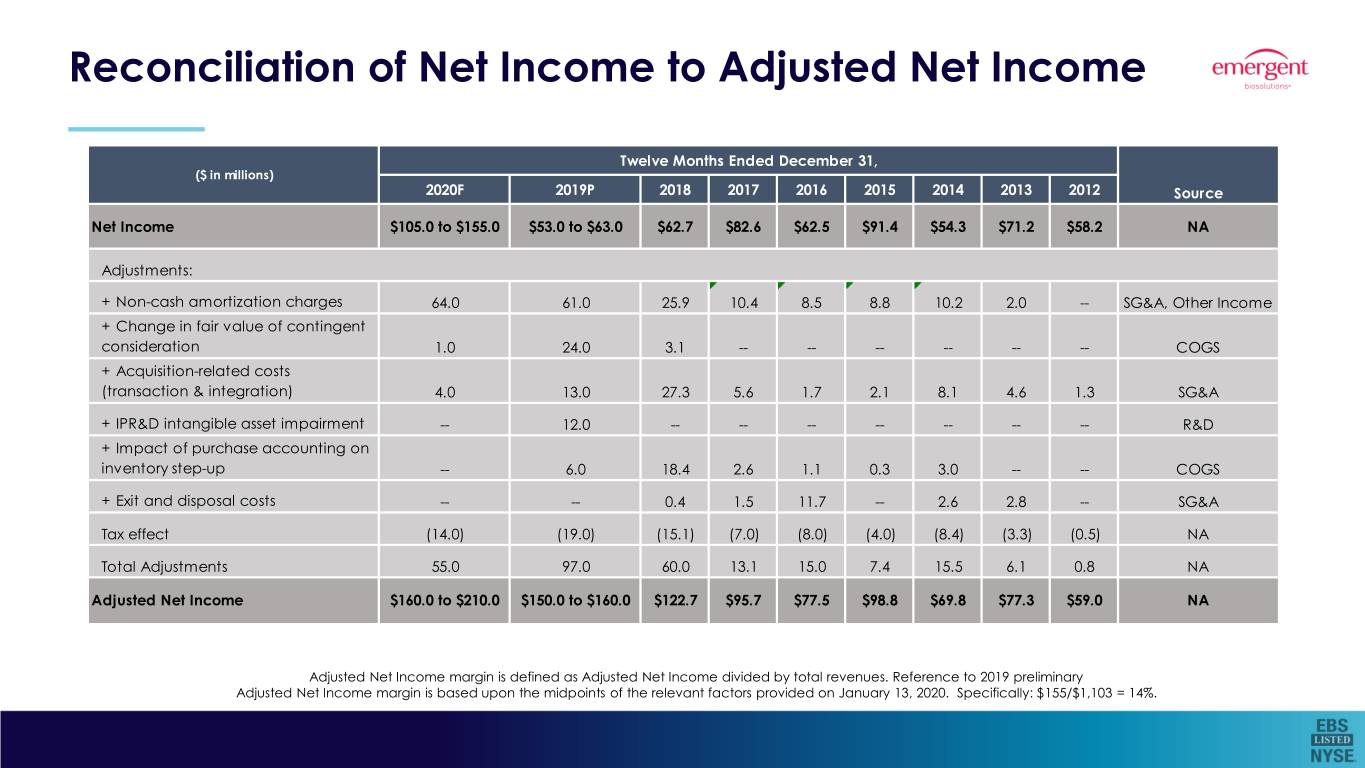
Reconciliation of Net Income to Adjusted Net Income Twelve Months Ended December 31, ($ in millions) 2020F 2019P 2018 2017 2016 2015 2014 2013 2012 Source Net Income $105.0 to $155.0 $53.0 to $63.0 $62.7 $82.6 $62.5 $91.4 $54.3 $71.2 $58.2 NA Adjustments: + Non-cash amortization charges 64.0 61.0 25.9 10.4 8.5 8.8 10.2 2.0 -- SG&A, Other Income + Change in fair value of contingent consideration 1.0 24.0 3.1 -- -- -- -- -- -- COGS + Acquisition-related costs (transaction & integration) 4.0 13.0 27.3 5.6 1.7 2.1 8.1 4.6 1.3 SG&A + IPR&D intangible asset impairment -- 12.0 -- -- -- -- -- -- -- R&D + Impact of purchase accounting on inventory step-up -- 6.0 18.4 2.6 1.1 0.3 3.0 -- -- COGS + Exit and disposal costs -- -- 0.4 1.5 11.7 -- 2.6 2.8 -- SG&A Tax effect (14.0) (19.0) (15.1) (7.0) (8.0) (4.0) (8.4) (3.3) (0.5) NA Total Adjustments 55.0 97.0 60.0 13.1 15.0 7.4 15.5 6.1 0.8 NA Adjusted Net Income $160.0 to $210.0 $150.0 to $160.0 $122.7 $95.7 $77.5 $98.8 $69.8 $77.3 $59.0 NA Adjusted Net Income margin is defined as Adjusted Net Income divided by total revenues. Reference to 2019 preliminary Adjusted Net Income margin is based upon the midpoints of the relevant factors provided on January 13, 2020. Specifically: $155/$1,103 = 14%.
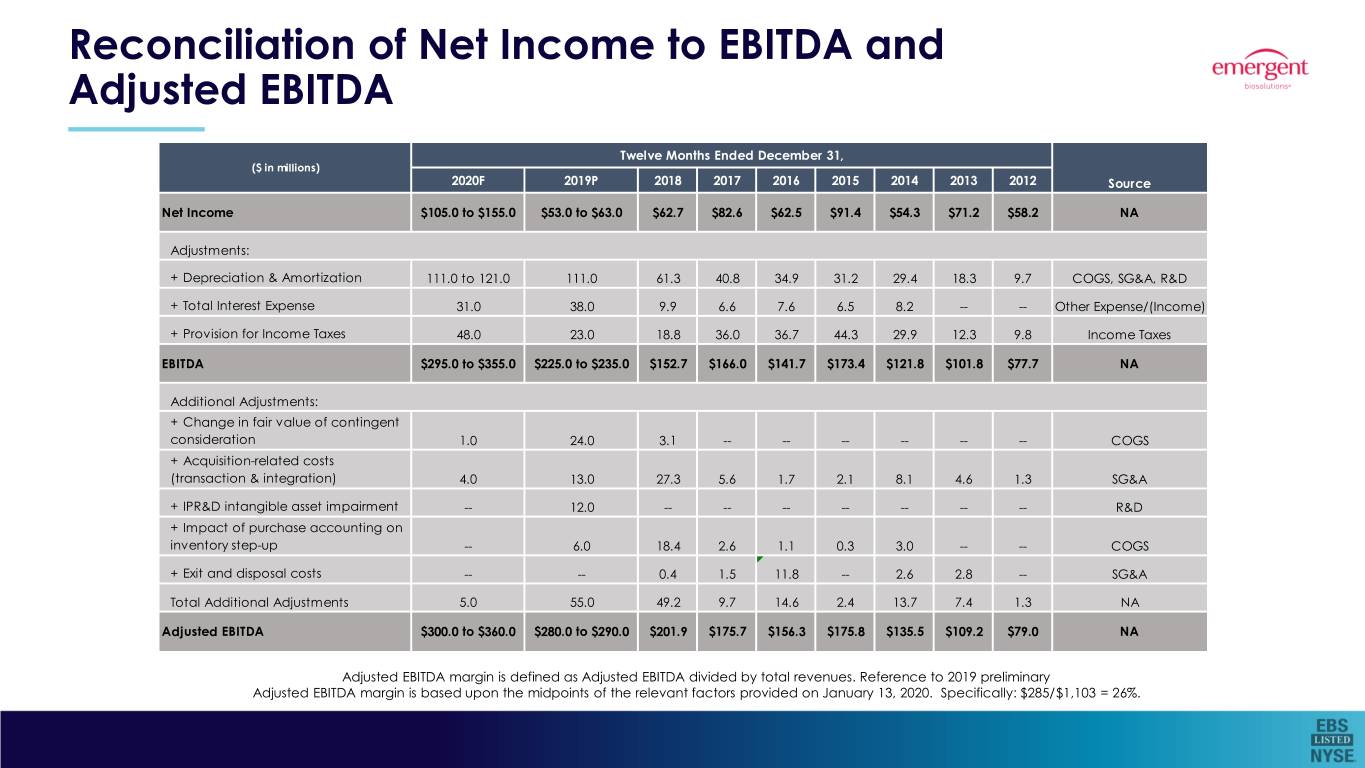
Reconciliation of Net Income to EBITDA and Adjusted EBITDA Twelve Months Ended December 31, ($ in millions) 2020F 2019P 2018 2017 2016 2015 2014 2013 2012 Source Net Income $105.0 to $155.0 $53.0 to $63.0 $62.7 $82.6 $62.5 $91.4 $54.3 $71.2 $58.2 NA Adjustments: + Depreciation & Amortization 111.0 to 121.0 111.0 61.3 40.8 34.9 31.2 29.4 18.3 9.7 COGS, SG&A, R&D + Total Interest Expense 31.0 38.0 9.9 6.6 7.6 6.5 8.2 -- -- Other Expense/(Income) + Provision for Income Taxes 48.0 23.0 18.8 36.0 36.7 44.3 29.9 12.3 9.8 Income Taxes EBITDA $295.0 to $355.0 $225.0 to $235.0 $152.7 $166.0 $141.7 $173.4 $121.8 $101.8 $77.7 NA Additional Adjustments: + Change in fair value of contingent consideration 1.0 24.0 3.1 -- -- -- -- -- -- COGS + Acquisition-related costs (transaction & integration) 4.0 13.0 27.3 5.6 1.7 2.1 8.1 4.6 1.3 SG&A + IPR&D intangible asset impairment -- 12.0 -- -- -- -- -- -- -- R&D + Impact of purchase accounting on inventory step-up -- 6.0 18.4 2.6 1.1 0.3 3.0 -- -- COGS + Exit and disposal costs -- -- 0.4 1.5 11.8 -- 2.6 2.8 -- SG&A Total Additional Adjustments 5.0 55.0 49.2 9.7 14.6 2.4 13.7 7.4 1.3 NA Adjusted EBITDA $300.0 to $360.0 $280.0 to $290.0 $201.9 $175.7 $156.3 $175.8 $135.5 $109.2 $79.0 NA Adjusted EBITDA margin is defined as Adjusted EBITDA divided by total revenues. Reference to 2019 preliminary Adjusted EBITDA margin is based upon the midpoints of the relevant factors provided on January 13, 2020. Specifically: $285/$1,103 = 26%.
